Abstract
Background: Total body irradiation (TBI) has long been incorporated as part of the conditioning regimen prior to hematopoietic stem cell transplant (HSCT). While the myeloablative TBI conditioning is associated with a lower relapse rate in high risk diseases such as Acute Myeloid Leukemia (AML), Myelodysplastic Syndrome (MDS), and Acute Lymphoblastic Leukemia (ALL), it is also associated with substantial toxicities, and increased NRM so use of this regimen is limited to young patients with excellent performance status. In this study, we used a linac-based volumetric modulated arc therapy (VMAT) technique to deliver standard myeloablative radiation to high risk body sites while sparing radiation sensitive organs (Organ Sparing Marrow Targeted Irradiation, OSMI). We hypothesized that this technique would be feasible and safe in patients who are older or who have higher transplant specific comorbidity index (HCT-CI), typically ineligible for standard TBI conditioning.
Methods: This is a single-arm prospective study. Patients from age 18-75 with high risk AML, MDS or ALL were eligible. There are 3 cohorts: (1) age 18-50 with HCT-CI of 3/4; (2) age 51-65 with HCT-CI of ≤ 3; and (3) age 66-75 with HCT-CI of ≤ 2. Patients receive OSMI to a total dose of 1200 cGy delivered twice daily for 6 fractions for a total of 7200 cGy. Clinical tumor volume includes total skeletal bone marrow and any sanctuary or high-risk areas. Graft-versus-host disease (GVHD) prophylaxis originally was tacrolimus and methotrexate. Given high incidence of bacterial infections related to mucositis, prophylaxis was changed to tacrolimus and sirolimus without methotrexate. All patients received Keratinocyte growth factor for prevention of mucositis. The primary objective was to assess feasibility and tolerability of OSMI based HSCT as defined by transplant-related mortality (TRM) at day 30 (D30) as well as rate of grade II/III organ toxicity (defined by Bearman Regimen-Related Toxicities Scale) attributable to conditioning occurring within 30 days.
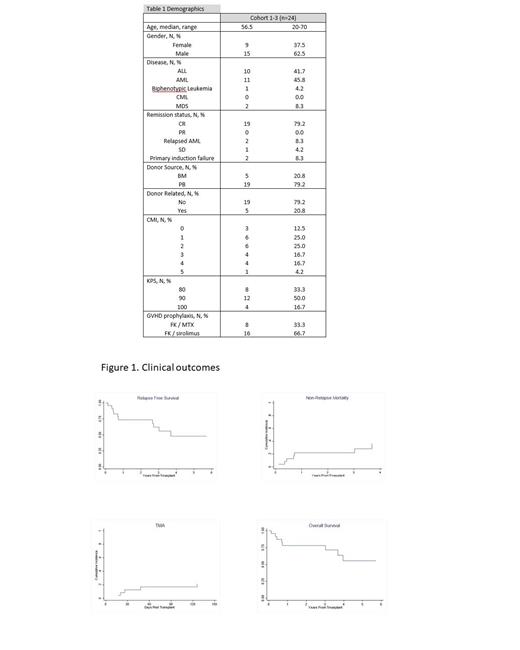
Results: Patient demographics for the 24 patients are shown in Table 1. Median age of recipients was 56.5 years. No graft failures were observed. The most common grade II or III Bearman toxicities include mucositis (grade 2: n=4), and diarrhea (grade 2: n=4). Clinical outcomes are shown in Figure 2. With a median follow-up of 3.3 years, overall survival (OS) and relapse-free survival (RFS) at 2 years was 78% and 74% respectively. Among the 5 patients who were not in complete remission at the time of transplant, 2-year OS and RFS rate was 40%. Incidence of Grades II-IV acute GVHD was 79% and Grades III-IV GVHD was 30%. Relapse incidence was 4% at 2 years. Incidence of Thrombotic microangiopathy by day 100 (TMA) as defined by Jodele's criteria was 17%. Incidence of chronic GVHD was 45% and severe chronic GVHD was 16%. One year non-relapse mortality was 22%, likely due to higher incidence of GVHD.
Conclusions: Selected patients who are older or with higher HCT-CI, who are typically not candidates for standard TBI conditioning, were able to receive a radiation-based myeloablative conditioning regimen with 2 year overall survival rates of 78%. We observed a high incidence of TMA, possibly related to use of tacrolimus and sirolimus as GVHD prophylaxis, and a high incidence of Grade II-IV acute GVHD. Low incidence of relapse was observed. OSMI-based conditioning was feasible in this cohort with median age of 56 years and was associated with low rates of relapse and favorable 2 year overall survival.
Vasu: Boehringer Ingelheim: Other: Travel support; Seattle Genetics: Other: travel support; Kiadis, Inc.: Research Funding; Omeros, Inc.: Membership on an entity's Board of Directors or advisory committees. Jaglowski: Novartis: Consultancy, Research Funding; Takeda: Consultancy; Juno: Consultancy; Kite, a Gilead Company: Consultancy, Research Funding; CRISPR Therapeutics: Consultancy. Brammer: Seattle Genetics: Speakers Bureau; Celgene: Research Funding; Kymera Therapeutics: Consultancy. de Lima: Incyte: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees; Miltenyi Biotec: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal